Boiler feed water treatment system is the lifeline of any thermal power plant or industrial steam generator. Whether the boiler is high-pressure, medium-pressure, or low-pressure, its performance and reliability depend heavily on how pure and well-conditioned the feed water is. Even minute quantities of dissolved solids, hardness, oxygen, silica, or contaminants can create severe operational problems such as scale formation, corrosion, sludge deposition, foaming, and carryover.
Boiler feed water treatment ensures that the water entering the boiler meets the strict chemical and physical standards required for safe and efficient boiler operation.
Impurities in Boiler Feed Water
Boiler feed water coming from raw water sources, pretreated water, or condensate return contains a variety of impurities. These contaminants directly influence scale formation, corrosion, and boiler water stability.
Suspended Solids
These include silt, sand, clay particles, and corrosion products. Suspended solids lead to sludge deposition inside the boiler, especially in low-velocity regions and tube bends.
Dissolved Solids
These ions remain dissolved in water and contribute to scaling, carryover, and deposits on boiler metal.
Common dissolved solids:
- Calcium (Ca²⁺)
- Magnesium (Mg²⁺)
- Sodium (Na⁺)
- Silica (SiO₂)
- Sulphates (SO₄²⁻)
- Chlorides (Cl⁻)
- Bicarbonates (HCO₃⁻)
Among these, calcium, magnesium, and silica are the major contributors to hard, adherent scale.
Dissolved Gases
The main dissolved gases are:
- Oxygen (O₂) – causes pitting and severe corrosion
- Carbon dioxide (CO₂) – forms carbonic acid in condensate lines
- Ammonia (NH₃) – attacks copper alloys
Even very small amounts of oxygen or carbon dioxide can rapidly corrode feedwater lines, economizers, and condensate systems.
Organic Impurities
Organic compounds from surface water or biological activity cause:
- Foaming
- Unstable boiler water
- Carryover of moisture with steam
Microbiological Impurities
Bacteria and algae may not survive boiler temperatures but can affect external treatment units (UF/RO) and create fouling in cooling and return systems.
Boiler Feed Water Treatment Overview
Boiler feed water treatment is the combined application of mechanical, chemical, and monitoring methods to ensure water entering the boiler is free from harmful impurities. Since modern high-pressure boilers generate steam at extremely high temperatures, even the smallest contaminants can cause scaling, corrosion, or carryover.
A complete feed water treatment system usually includes:
✔ External Treatment – Removes impurities before water enters the boiler.
✔ Internal Treatment – Controls chemistry inside the boiler using chemicals.
✔ Condensate Treatment – Maintains purity in the steam return line.
A balanced combination of these ensures:
- Stable boiler operation
- Minimal blowdown
- Reduced chemical consumption
- High steam purity (important for turbines)
- Longer boiler life
✅ External Treatment (Before water enters the boiler)
External treatment focuses on removing suspended solids, dissolved solids, hardness, dissolved gases, and alkalinity from raw water or makeup water.
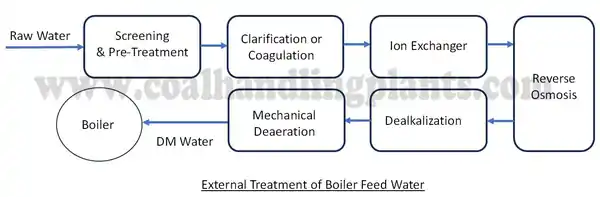
1. Screening & Pre-Treatment
Raw water often contains large impurities like leaves, debris, sand, and coarse solids. These are removed through:
- Screens
- Strainers
- Grit chambers
This protects downstream equipment.
2. Clarification / Coagulation
Chemicals like alum or polyaluminum chloride are added to remove fine particles by forming flocs. This process reduces turbidity, color, and suspended solids.
Steps:
- Rapid mixing
- Floc formation
- Settling
- Sludge removal
This also decreases organic matter, reducing the chance of foaming later in the boiler.
3. Filtration & Ultrafiltration
Filtration types:
- Pressure sand filters
- Activated carbon filters
- Dual media filters
Ultrafiltration (UF)
UF membranes remove particles down to 0.01–0.1 microns, ensuring consistently low turbidity. It protects RO membranes and reduces fouling.
4. Ion Exchange (Softening / Demineralization)
Softening
Uses strong acid cation (SAC) resin to remove hardness (Ca²⁺, Mg²⁺) by exchanging them with Na⁺ ions.
Demineralization (DM)
For high-pressure boilers, hardness, alkalinity, silica, and salts must be nearly zero.
DM plant consists of:
- Cation exchanger
- Anion exchanger
- Mixed-bed exchanger
Produces high-purity water.
5. Reverse Osmosis (RO) / Nanofiltration (NF)
RO removes:
- 98% dissolved salts
- Silica
- Organics
- Heavy metals
- Alkalinity
RO is widely used because it significantly reduces the load on DM units.
6. Dealkalization
Removes alkalinity (HCO₃⁻, CO₃²⁻) that could otherwise cause:
- Foaming
- High boiler water alkalinity
- Caustic corrosion
Strong anion resin or weak acid cation resin is used.
7. Mechanical Deaeration
A deaerator removes oxygen and carbon dioxide by heating water to saturation temperature.
Advantages:
- Reduces O₂ below safe limits
- Prevents feed line and economizer corrosion
- Reduces chemical consumption
✅ Internal Treatment (Inside the boiler using chemicals)
Internal treatment is required to handle any impurities that remain after external treatment and to maintain stable boiler water chemistry.
1. Phosphate Conditioning
Phosphates convert hardness into soft, non-adherent sludge.
Types of phosphate programs:
- Conventional phosphate – Uses caustic along with phosphate
- Coordinated phosphate – Controls pH and phosphate together
- Congruent phosphate – Prevents hideout in high-pressure boilers
These methods prevent scale formation on heating surfaces.
2. Alkalinity / Caustic Control
Caustic (NaOH) is used to maintain boiler water alkalinity. Proper alkalinity prevents acidic corrosion and maintains protective magnetite film on tube surfaces.
3. Polymer / Dispersant Treatment
Polymers help:
- Keep precipitated solids dispersed
- Prevent hard scale
- Prevent sludge from settling
They enable easier blowdown removal.
4. Chelant Treatment (EDTA / NTA)
Chelants combine with hardness ions to keep them soluble. Used only in carefully controlled operating conditions because over-chelation can cause corrosion.
5. Organic Conditioning Agents
Natural or synthetic organics like tannins or lignins modify crystal structures and prevent scale formation.
6. Oxygen Scavengers
Used after the deaerator to remove residual oxygen.
Common scavengers:
- Hydrazine
- Sodium sulphite
- DEHA
- Carbohydrazide
These prevent oxygen pitting in economizers and feed lines.
7. Chemical Dosing Systems & Injection Equipment
Includes:
- Chemical day tanks
- Agitators
- Dosing pumps
- Injection quills
- Flow controllers
Continuous dosing is preferred to avoid fluctuations in boiler chemistry.
8. Blowdown Management
Blowdown removes concentrated boiler water.
Types:
- Continuous blowdown – Controls TDS
- Intermittent blowdown – Removes sludge
Correct blowdown reduces carryover and protects the boiler.
✅ Condensate Water Treatment (Treatment in the steam return/condensate lines)
Condensate is one of the purest forms of water in a boiler cycle, but it can get contaminated while returning from process equipment or turbine extractions. Protecting condensate quality is essential because any contamination directly enters the feed water circuit.
1. Neutralizing Amines
Neutralizing amines raise the pH of condensate by neutralizing carbonic acid (formed from CO₂).
Common neutralizing amines:
- Morpholine
- Cyclohexylamine
- Diethylaminoethanol (DEAE)
They prevent corrosion in long condensate lines.
2. Filming Amines
These amines form a protective, hydrophobic film on metal surfaces, preventing moisture and oxygen from causing corrosion.
Best suited for:
- Long steam distribution networks
- Return lines with varying pH
- Systems with oxygen ingress
3. Return Line Corrosion Control
Corrosion in return lines is mainly caused by:
- CO₂ → carbonic acid
- Dissolved oxygen
- Low pH
- Iron/copper dissolution
Treatment measures include:
- Maintaining correct condensate pH
- Using neutralizing & filming amines
- Fixing vacuum leaks
- Ensuring deaerator performance
4. Condensate Polishing Units (CPU)
CPU removes:
- Iron
- Copper
- Silica
- Oil contamination
Polished condensate significantly reduces boiler blowdown and chemical usage.
5. Monitoring Iron & Copper Levels
High iron indicates feed line or boiler corrosion. High copper indicates condensate system corrosion.
Maintaining low Fe/Cu in condensate ensures safe boiler operation.
Monitoring & Control Parameters
A reliable feed water and boiler water treatment program requires frequent monitoring of key parameters.
1. pH
Controls corrosion and deposit formation. Ideal ranges vary for feed water, boiler water, and condensate.
2. Conductivity
Indicates dissolved solids concentration. Used for automatic blowdown control.
3. Total Dissolved Solids (TDS)
High TDS → foaming & carryover
Low TDS → safe operation
4. Hardness
Should be nearly zero. Prevents hardness scale.
5. Silica
Critical for high-pressure boilers.
Silica carryover → turbine blade deposits.
6. Dissolved Oxygen
Must be minimized to prevent pitting.
Mechanical deaeration + scavengers ensure acceptable limits.
7. Phosphate Concentration
Maintained within specific ranges depending on treatment method.
8. Iron / Copper
Indicate corrosion levels in return lines, feed system, or boiler.
Problems & Troubleshooting of Boiler Feed Water Treatment System
1. Scale Formation
Cause: Hardness ions (Calcium & Magnesium), Silica, and high boiler water concentration form hard deposits on tube surfaces.
Effect: Higher fuel consumption, overheating, tube bulging and failure.
Solution: Use softener, RO, or DM plant, Maintain correct phosphate & alkalinity levels., Use polymers/dispersants., Control TDS through proper blowdown.
2. Sludge Deposition
Cause: Suspended solids, magnesium salts, corrosion products, and improper blowdown settle in low-flow areas.
Effect: Flow blockage, reduced heat transfer, boiler instability.
Solution: Improve filtration/UF, Use polymers to keep sludge suspended, Perform regular bottom blowdown, Increase feed water quality.
3. Corrosion
Cause: Presence of oxygen, CO₂, low pH, acidic contamination, or high dissolved salts.
Effect: Pitting corrosion, economizer damage, feed line leaks, condensate corrosion.
Solution: Ensure proper deaerator performance, Dose oxygen scavengers (hydrazine, sulphite, DEHA), Maintain correct pH/alkalinity, Use neutralizing or filming amines in condensate.
4. Foaming & Carryover
Cause: High TDS, high alkalinity, organic matter, and oil contamination.
Effect: Water droplets enter steam → turbine deposits, wet steam, loss of steam purity.
Solution: Control TDS via continuous blowdown, Reduce alkalinity, Remove organics through pretreatment, Use anti-foam if required.
5. Energy Loss
Cause: Scale, sludge, and corrosion products reduce heat transfer efficiency.
Effect: Increased fuel consumption, reduced steam generation.
Solution: Maintain clean heat transfer surfaces, Prevent scale/sludge through proper treatment, Optimize blowdown to maintain safe TDS.
6. Tube Failures
Cause: Combination of scale, corrosion, overheating, improper water chemistry control.
Effect: Tube rupture, boiler trips, unplanned shutdowns.
Solution: Maintain proper feed water quality, Ensure chemical dosing accuracy, Prevent overheating by controlling scale, Conduct periodic tube inspections and cleaning.
Best Practices in Feed Water Treatment
- Test feed water, boiler water & condensate daily
- Use continuous dosing instead of intermittent
- Maintain deaerator temperature close to saturation
- Keep blowdown within recommended limits
- Monitor iron/copper to detect corrosion early
- Maintain correct phosphate/alkalinity ratio
- Train operators on reading boiler chemistry trends
- Fix leaks in condensate system to reduce makeup water
- Clean strainers and filters regularly
- Ensure spare dosing pumps are always functional
Frequently Asked Questions (FAQs) on Boiler Feed Water Treatment System
To prevent scale, corrosion, carryover, energy loss, and boiler damage.
External treatment purifies water before boiler entry; internal treatment controls water chemistry inside the boiler.
Silica volatilizes with steam and forms hard glassy deposits on turbine blades.
Phosphate temporarily hides in deposits under high load and reappears when load falls.
Phosphate, caustic soda, polymers, oxygen scavengers, amines.
Only if properly polished and free from oil, iron, copper and organics.
Typically alkaline (exact values vary with boiler pressure).
Due to oxygen pitting caused by inadequate deaeration.
Depends on TDS; typically 2–10% for most boilers.
No. Internal chemical treatment is still required.
